Heart Failure and SGLT2 Inhibitors: Access Has Improved, Utilization Has Not.
Why the GDMT Gap in Heart Failure Needs a Different Playbook in 2026
For most of the last five years, the dominant explanation for why so few heart failure patients receive guideline-directed medical therapy has been access. Formulary restrictions. Prior authorization friction. Step therapy requirements. Patient out-of-pocket exposure. The argument has been that if we could fix the access architecture, utilization would follow.
The access architecture has now shifted. The Inflation Reduction Act's (IRA) Medicare Drug Price Negotiation Program took effect January 1, 2026. Jardiance is now $197 per 30-day supply, down from $573. Farxiga is $178.50, down from $556. Entresto is also on the list. The Part D redesign is fully maturing, with a $2,100 annual out-of-pocket cap, no coverage gap, and a smoothed payment option available to all enrollees. Generic dapagliflozin has been available since 2024. Kazi and colleagues (2023) projected that the IRA would reduce annual out-of-pocket costs for HFrEF patients by approximately $804, and by more than $1,200 for patients with HFrEF and atrial fibrillation. Trish et al. (2024) documented that pre-IRA, Medicare patients on these agents faced meaningful coverage-gap exposure mid-year, with associated discontinuation.
Cost was never irrelevant, but it mattered. It still matters for commercial and Medicaid populations, which did not get the IRA reset. But the evidence base from before the IRA already told us something important: that access and cost were contributors to the utilization gap, but perhaps not the dominant drivers. The 2026 reset for Medicare simply makes this harder to ignore.

What The Evidence Actually Shows
The AHA’s Get With The Guidelines Heart Failure registry analysis by Pierce, Greene, and colleagues (JAMA Cardiology, 2023) remains the cleanest look at prescribing behavior. Among 49,399 patients hospitalized for heart failure with reduced ejection fraction (HFrEF) between July 2021 and June 2022, SGLT2 inhibitor (SGLT2i) prescription at discharge was approximately 20 percent among eligible patients. Only 4.1 percent of participating hospitals discharged half or more of their eligible patients on an SGLT2i.
That finding is important, but the more revealing finding is the variation. Some hospitals discharged 100 percent of eligible patients on an SGLT2i; others discharged zero. The median odds ratio for between-hospital variation was 2.51 and persisted after adjustment for patient and hospital characteristics, including insurance status. The same evidence base, same clinical guidelines, same class 1A recommendation, yet dramatically different outcomes patient to patient, based primarily on which hospital the patient happened to be admitted to.
The Veterans Affairs analysis by Hussain, Mahtta, and colleagues of 105,799 patients with atherosclerotic cardiovascular disease, heart failure, and type 2 diabetes showed 14.6 percent SGLT2i utilization across 130 facilities, with a median rate ratio of 1.55 between similar facilities. The Shin et al. JACC analysis (2024) across 28 U.S. health systems showed prescription rates of only about 12 percent among patients with diabetes and a class 1A recommendation, with no health system exceeding roughly 25 percent.
The gap is also narrowing, which matters for how we frame the next two years. Abdel Jawad et al. (2025) documented that SGLT2i prescription for HFmrEF and HFpEF increased from 4.2 percent in Q3 2021 to 23.5 percent in Q3 2023. El Rafei and colleagues (2025) showed that in ambulatory cardiovascular care, prescribing rose from 4.6 percent in Q3 2019 to 16.2 percent in Q2 2023 across all heart failure patients, and from 5.1 percent to 28.5 percent in HFrEF specifically. The trajectory is correct. The magnitude still represents a substantial unmet clinical need, and practice-level variation in the El Rafei data was a 4.4-fold difference between 90th and 10th percentile sites after adjustment.
Taken together, the evidence suggests that prescribing variation is not primarily a function of who pays. It is a function of how care is organized, how decisions are made, and how eligible patients are identified in the first place.
The 2026 Reset Changes The “Diagnosis”
When you remove a meaningful portion of the cost and formulary friction from the equation, as the IRA has done for Medicare patients on the negotiated agents, what remains? Several things, and they are more actionable than the access story was. The contribution estimates below are informed synthesis across the literature rather than precise measurements, and they overlap. They are offered as a framework for prioritization, but not as a definitive assessment in the complex environment.
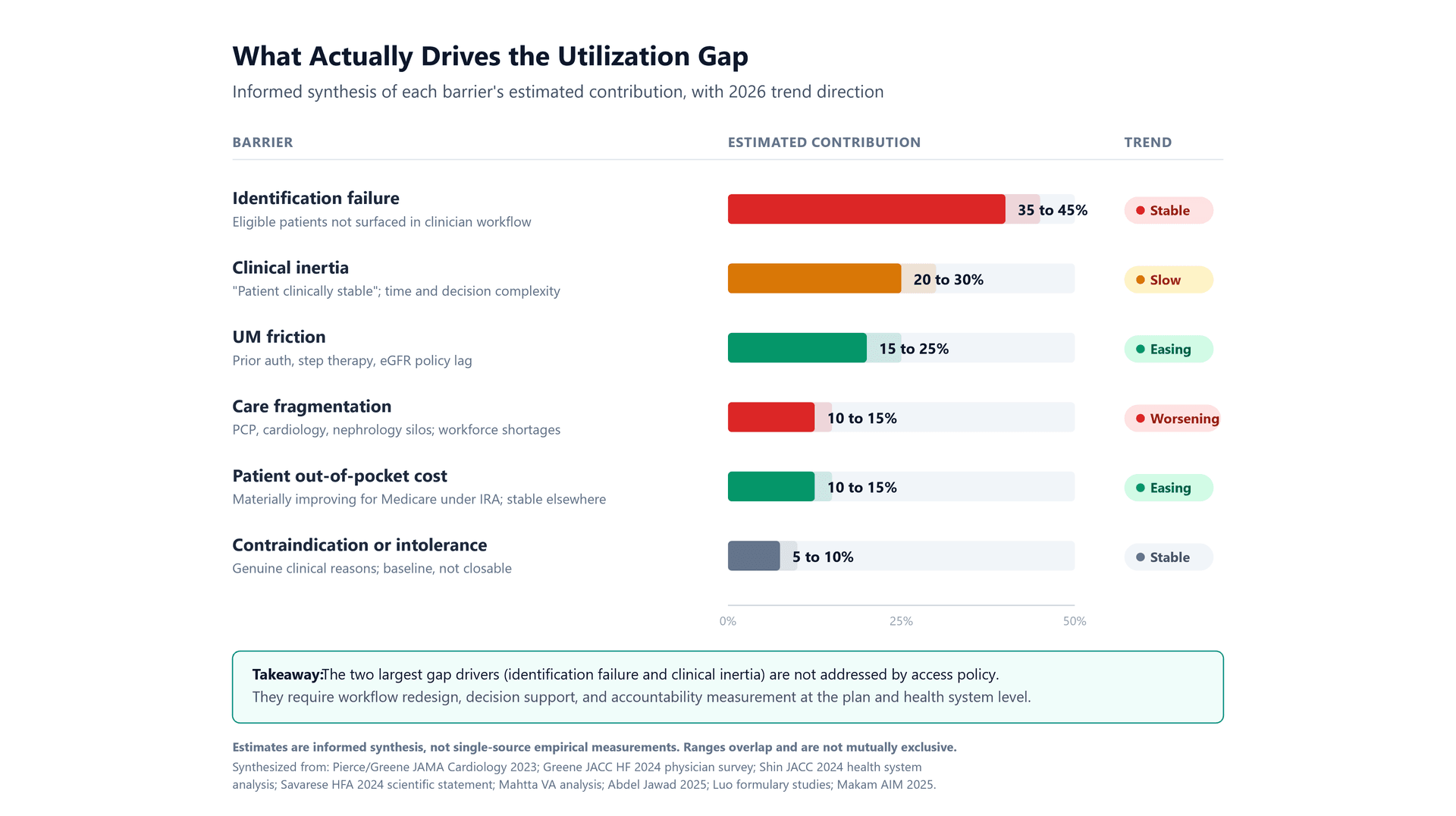
- Identification failure (estimated 35 to 45 percent of the gap). In most primary care and cardiology workflows, the question of whether a patient is eligible for GDMT initiation is answered episodically, during an office visit, using whatever data is visible in the EHR in that moment. Patients with heart failure who are not coded correctly, whose ejection fraction data lives in an unstructured note, whose albuminuria was last measured eighteen months ago, or whose diabetes status is controlled well enough to be deprioritized clinically, do not get flagged. The Shin et al. finding that even health systems with structured data top out around 24 percent suggests identification is a substantial contributor, even where information infrastructure is strong.
- Clinical inertia (estimated 20 to 30 percent). Greene and colleagues (JACC Heart Failure, 2024) directly surveyed physicians on reasons for not initiating GDMT and found that the dominant reason across medication classes was that patients were "clinically stable and/or had adequate symptom control," accounting for roughly half of all non-prescriptions. This is inertia in its most consequential form. The patient looks fine. Nothing seems urgent. The prescriber moves on. The Savarese et al. 2024 HFA survey adds organizational factors, time constraints, and the complexity of managing four-pillar therapy simultaneously as additional inertia drivers. This is not primarily a knowledge problem. It is a decision-making and workflow problem.
- Care fragmentation (estimated 10 to 15 percent). A patient with heart failure, chronic kidney disease, type 2 diabetes, and atherosclerotic cardiovascular disease is being seen by a primary care physician, a cardiologist, a nephrologist, and sometimes an endocrinologist. Each specialist owns a piece of the GDMT picture. The pharmacist is often the only member of the care team who sees all four medication classes at once, but the pharmacists are structurally disadvantaged from initiating therapy in most practice settings.
- Utilization management friction that has not yet caught up to the label (estimated 15 to 25 percent, easing for Medicare in 2026). Empagliflozin can be initiated for heart failure and chronic kidney disease down to an eGFR of 20 mL/min and continued below that threshold once established. Dapagliflozin carries a similar initiation threshold eGFR of 25 mL/min for chronic kidney disease. Many plan medical policies still reflect older eGFR thresholds of 30 or 45 mL/min. Some plans still require documented failure or trial of an ACE inhibitor, ARB, or ARNi before approving an SGLT2i, which conflicts with the current guideline preference for parallel initiation of the four pillars. These are solvable problems, but they require plan-level policy refresh.
Why PQA's Heart Failure Medication Quality Measure Development Matters
On March 4, 2026, the Pharmacy Quality Alliance announced a new measure development technical expert panel to build a health plan measure titled Evidence of Guideline-Directed Medical Therapy in Persons with Heart Failure. The measure will assess the percentage of individuals with prescription claims for guideline-directed medical therapies for heart failure, covering all four pillars: 1) ARNi, ACEi, or ARB, 2) beta-blockers, 3) mineralocorticoid receptor antagonists (MRAs); and 4) SGLT2i’s. The TEP scheduled to hold its first meeting on April 22, 2026.

This is a meaningful development, and it is the piece of the story that has been missing. Quality measurement is how accountability gets attached to prescribing behavior at the health plan level. Measures drive Stars ratings. Stars ratings drive Medicare Advantage rebate dollars. Rebate dollars drive plan investment in clinical programs. Clinical programs drive member outreach, provider engagement, and care pathway redesign.
The path from PQA measure development to Stars adoption is typically a three-to-five year arc, with testing, endorsement, and rulemaking between measure specification and program incorporation with CMS. This measure may not reach Stars in its first iteration, or in its current form. But PQA measure development is the signal that the accountability architecture for heart failure GDMT is being built, and health plans, provider groups, and life sciences companies that anticipate this environment will be better positioned than those who wait for it to arrive. Quite the opportunity for the ACC and AHA to also elevate the impact of science on healthcare economic value.
Without a plan-level measure for GDMT in heart failure, the utilization gap is a clinical problem that plans can observe but not be held accountable for. With one, the gap becomes a financial performance issue. That changes the conversation quickly.
The measure also creates a natural forcing function for the workflow changes that actually close the gap. Plans that want to perform well on a GDMT measure will need to identify their heart failure population accurately, engage prescribers proactively, flag eligible patients who are not on therapy, and reduce internal utilization management friction on their own formulary. All of those activities are upstream of the individual prescribing decision, and all of them are where the gap actually lives.
What This Means For Each Stakeholder In 2026
- For health plans and pharmacy benefit managers: The access story is ending. The accountability story is beginning. Plans that have eGFR thresholds below current initiation labeling, still require sequential rather than parallel GDMT initiation, or still default to ertugliflozin for formulary preference despite its lack of heart failure and chronic kidney disease indications, will have real medical policy work to do ahead of the coming measurement environment.
- For health systems and provider groups: The Duke and GWTG-HF variation data is the single most important number in your quality strategy for the next two years. If some peer hospitals can discharge 100 percent of eligible heart failure patients on an SGLT2i and yours discharges 20 percent, that is not a patient mix problem. That is a workflow problem. Identification, decision support, and prescriber engagement at the point of discharge are the levers.
- For life sciences and health technology companies: The commercial argument is shifting. "We solve an access problem" is a weaker pitch in 2026 than it was in 2024. "We solve an identification and workflow problem that plans are about to be measured on" is a stronger pitch. Any technology that can accurately identify eligible heart failure patients at scale, surface them into a clinician workflow, and support initiation decisions has a much clearer buyer and a much clearer ROI than it did a year ago.
- For patients and caregivers: The 2026 Medicare landscape is genuinely better. A patient on Jardiance or Farxiga under Part D now has lower negotiated pricing, a $2,100 out-of-pocket cap, and the option to smooth payments across the year. Commercial and Medicaid patients still face meaningful variation, and coverage-gap behavior patterns documented by Trish and colleagues will take time to fully resolve. Anyone with a heart failure diagnosis should have an active conversation with their prescriber about whether they are on all four pillars of GDMT, and if not, why not.
Conclusion
The SGLT2i utilization gap has always been a composite problem. Access was a real contributor, but the pre-IRA evidence base already told us it was not the dominant one. The 2026 IRA reset removes enough of the access friction for the Medicare population to make this visible in real time. What else is going on is identification failure, stability-anchored clinical inertia, care fragmentation, and utilization management policy that has not kept pace with the evidence.
PQA's measure development is the right intervention at the right time. If the TEP produces a workable plan-level GDMT measure, and if CMS eventually adopts it into Stars, the accountability architecture for heart failure quality will finally match the clinical evidence base. That is the shift that turns an underutilization problem into a solved problem.
Until then, the work is ours. Plans need to refresh medical policy. Health systems need to address workflow variation. Life sciences and health technology need to build for identification and decision support, not just access. And patients need a care team that treats all four pillars of GDMT as a package, not a sequence.
--------------------------------------------------------------
Erik Abel, PharmD, MBA, is Founder and Principal Healthcare Strategist of OneAnother Health, LLC. He writes about payer strategy, value-based care, and health technology commercialization at erikabel.org.
Key References
- Pierce JB, Vaduganathan M, Fonarow GC, et al. Contemporary Use of Sodium-Glucose Cotransporter-2 Inhibitor Therapy Among Patients Hospitalized for Heart Failure With Reduced Ejection Fraction in the US: The Get With The Guidelines-Heart Failure Registry. JAMA Cardiol. 2023;8(7):652-661.
- Hussain A, Ramsey D, Lee M, Mahtta D, et al. Utilization Rates of SGLT2 Inhibitors Among Patients With Type 2 Diabetes, Heart Failure, and Atherosclerotic Cardiovascular Disease: Insights From the Department of Veterans Affairs. JACC Heart Fail. 2023.
- Shin JI, Xu Y, Chang AR, et al. Prescription Patterns for Sodium-Glucose Cotransporter 2 Inhibitors in U.S. Health Systems. J Am Coll Cardiol. 2024;84(8):683-693.
- Greene SJ, Bash LD, Tebbs KW, et al. Physician-Reported Reasons for Not Initiating Guideline-Directed Medical Therapy for Heart Failure. JACC Heart Fail. 2024;12(12):2120-2122.
- Savarese G, Lindberg F, Christodorescu RM, et al. Physician perceptions, attitudes, and strategies towards implementing guideline-directed medical therapy in heart failure with reduced ejection fraction. A survey of the Heart Failure Association of the ESC and the ESC Council for Cardiology Practice. Eur J Heart Fail. 2024;26(6):1408-1418.
- Abdel Jawad M, Spertus JA, Ikeaba U, et al. Early Adoption of Sodium-Glucose Cotransporter-2 Inhibitor in Patients Hospitalized With Heart Failure With Mildly Reduced or Preserved Ejection Fraction. JAMA Cardiol. 2025;10(1):89-94.
- El Rafei A, Gosch K, Manning ES, et al. Sodium-Glucose Cotransporter 2 Inhibitor Use for Heart Failure in US Ambulatory Cardiovascular Care. JAMA Cardiol. 2025;10(9):904-913.
- Kazi DS, DeJong C, Chen R, Wadhera RK, Tseng CW. The Inflation Reduction Act and Out-of-Pocket Drug Costs for Medicare Beneficiaries With Cardiovascular Disease. J Am Coll Cardiol. 2023;81(21):2103-2111.
- Trish E, et al. The Inflation Reduction Act and Patient Costs for Drugs to Treat Heart Failure. JAMA Health Forum. 2024.
- Pharmacy Quality Alliance. "PQA Is Launching a Heart Failure Technical Expert Panel." March 4, 2026.
