Biosimilars and Patient Out-of-Pocket Costs: The Hidden Affordability Problem
The $5 to $500 Problem: This Is Not a Biosimilar Problem. It Is a Benefit Design Problem.
Biosimilars were designed under the core premise of decreasing drug spending through competition.
In many payer analyses, they do. But for commercially insured patients, the experience at the pharmacy counter tells a different story. Lower net payer cost does not automatically translate into lower patient out-of-pocket (OOP) costs. In some cases, OOP exposure increases after biosimilar entry.
Worse yet, when affordability worsens, adherence falls...and when adherence falls, medical costs and utilization rises.
This is the patient cost paradox in the biosimilar era.
Humira vs Biosimilars: How Copay Assistance Supports Medication Adherence
Originator biologics such as adalimumab (Humira) have historically been paired with robust patient support programs (PSPs) and copay assistance. These programs significantly influence adherence and persistence.
A managed care analysis of HUMIRA’s patient support program found:
- 29% higher medication adherence (64.8% vs 50.1%)
- 22% lower discontinuation (51.4% vs 65.9%)
- 35% lower disease-related medical costs ($10,162 vs $15,511)
- Despite 12% higher drug spending, total healthcare costs were 9% lower among PSP participants.¹
Nationally, manufacturer copay cards offset an estimated $19.8 billion in patient OOP costs in 2023, with 63% of brand-name prescriptions using copay assistance.²
Lower point-of-sale cost improves persistence on high-value therapy. The data are consistent across chronic conditions.
Do Biosimilars Lower Patient Out-of-Pocket Costs? Evidence from Commercial Insurance
The assumption that biosimilar competition reduces patient affordability burden does not consistently hold true in commercial markets.
Recent analyses show:
- Biosimilar claims were 13% more likely to have nonzero OOP compared with reference biologics (adjusted OR 1.13).³
- Annual OOP spending increased 12% within two years after biosimilar availability.³
- Early infliximab biosimilar competition showed no meaningful improvement in patient OOP affordability.⁴
Biosimilars may reduce acquisition cost, but benefit design determines patient exposure.
Commercial reimbursement structures are rebate-driven and contract-specific. Coinsurance, deductibles, tier placement, and accumulator policies can offset or negate acquisition savings at the patient level.
The result can be a cost cliff rather than cost relief.
Copay Accumulator Adjustment Programs (CAAPs): Impact on Adherence and Discontinuation
Copay accumulator adjustment programs prevent manufacturer assistance from counting toward deductibles or annual out-of-pocket maximums.
Their impact on specialty drug adherence is measurable:
- 233 fewer fills per 1,000 patients
- 20% increased discontinuation
- 12% lower adherence in high-deductible plans⁵
States that banned copay accumulators experienced:
- 41–63% reductions in patient liability
- 14% greater odds of adherence
- 13% reduction in discontinuation risk⁶
This is benefit design shaping patient behavior.
Autoimmune Medication Abandonment and High Out-of-Pocket Costs
The relationship between OOP cost and prescription abandonment in RA is steep. Among new initiators of biologic therapy:
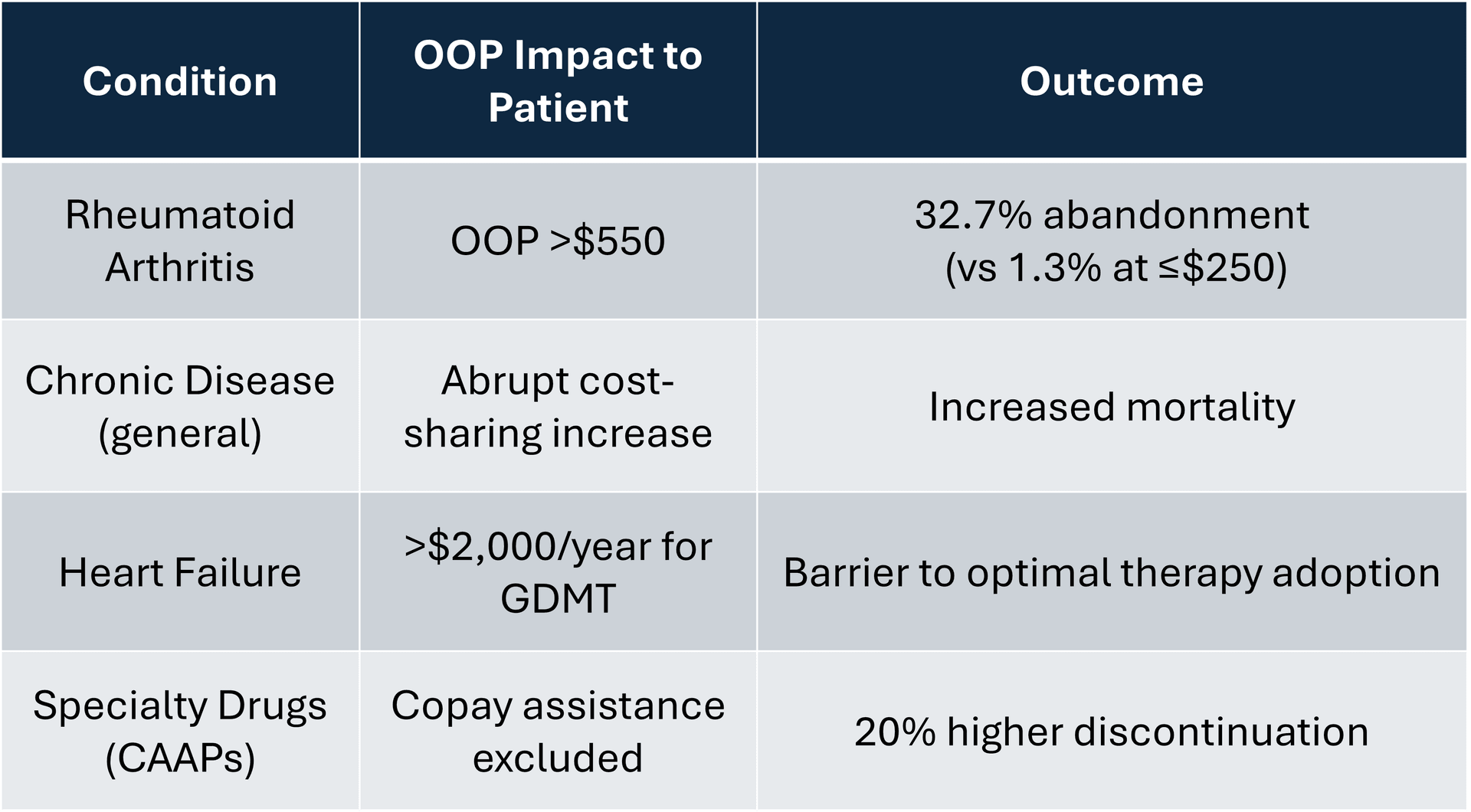
- Abandonment was 1.3% when OOP ≤ $250
- Abandonment rose to 32.7% when OOP > $550⁷
- Copay assistance was associated with 79% lower odds of abandonment.⁸
Patients with rheumatoid arthritis (RA) for instance have a 3.5-fold higher risk of cost-related medication non-adherence compared with individuals without chronic disease.⁹
When a stable patient paying $5 per month with copay support is switched to a biosimilar without equivalent assistance AND OOP increases to several hundred dollars per fill, abandonment and discontinuation risk rise sharply.
Net savings at the plan level do not eliminate financial toxicity at the patient level.
Medicare vs Commercial Insurance: Why Biosimilar Out-of-Pocket Trends Differ
Medicare beneficiaries experience different dynamics. A 2026 study found biosimilar competition was associated with lower OOP costs in Medicare, with mean annual patient spending declining from over $200 to approximately $165 within four years.¹⁰
The structural reason is reimbursement design. Medicare reimbursement is directly linked to average sales price (ASP). Commercial markets operate within rebate-based frameworks and variable benefit structures that can diverge from acquisition cost trends. This divergence explains why biosimilar affordability improvements appear more consistently in Medicare than in commercial plans.
Cost Sharing, Medication Adherence, and Mortality: Broader Evidence Across Chronic Disease
The RA findings align with broader research on cost sharing and health outcomes. A National Bureau of Economic Research study examining abrupt increases in cost sharing found that higher OOP exposure reduced medication use and increased mortality, even for high-value therapies.¹¹
In heart failure, Medicare Part D benefit design has been associated with median annual OOP costs exceeding $2,000 for guideline-directed quadruple therapy (GDMT), creating barriers to optimal treatment adoption.¹²
Across disease states, the pattern is consistent as shown below:
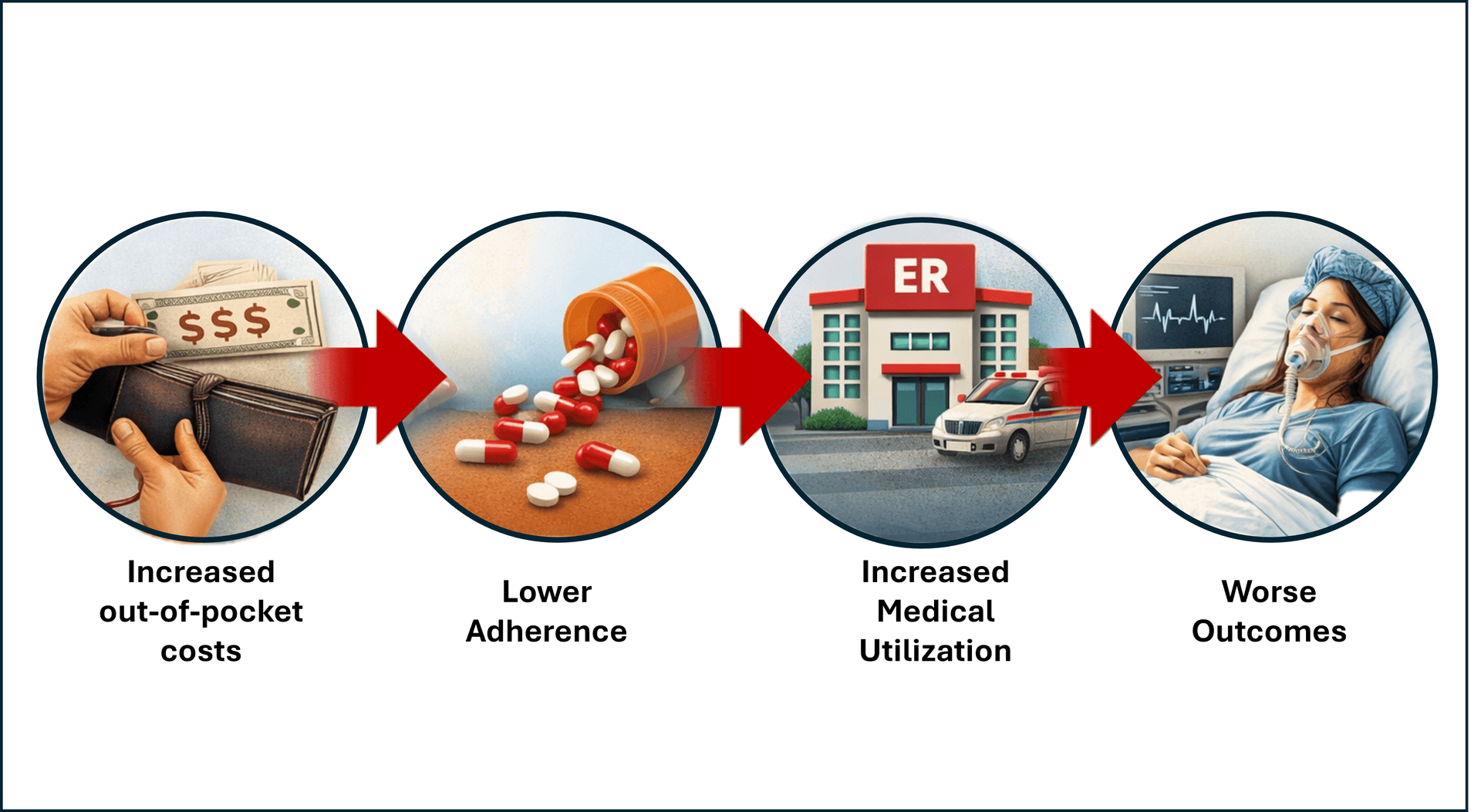
At the same time, many health plans report persistent concerns about rising utilization patterns and avoidable acute care spend.
It may be worth examining whether benefit design itself is contributing to that utilization. When high-value chronic therapies are exposed to abrupt or unpredictable cost sharing, reduced adherence should not be surprising. In that context, some portion of downstream utilization may not be a failure of patient behavior, but rather a predictable response to OOP financial exposure.
If affordability drives adherence, and adherence drives outcomes, then benefit design becomes part of the causal pathway.
The Biosimilar Cost Paradox: Net Price vs Patient Price
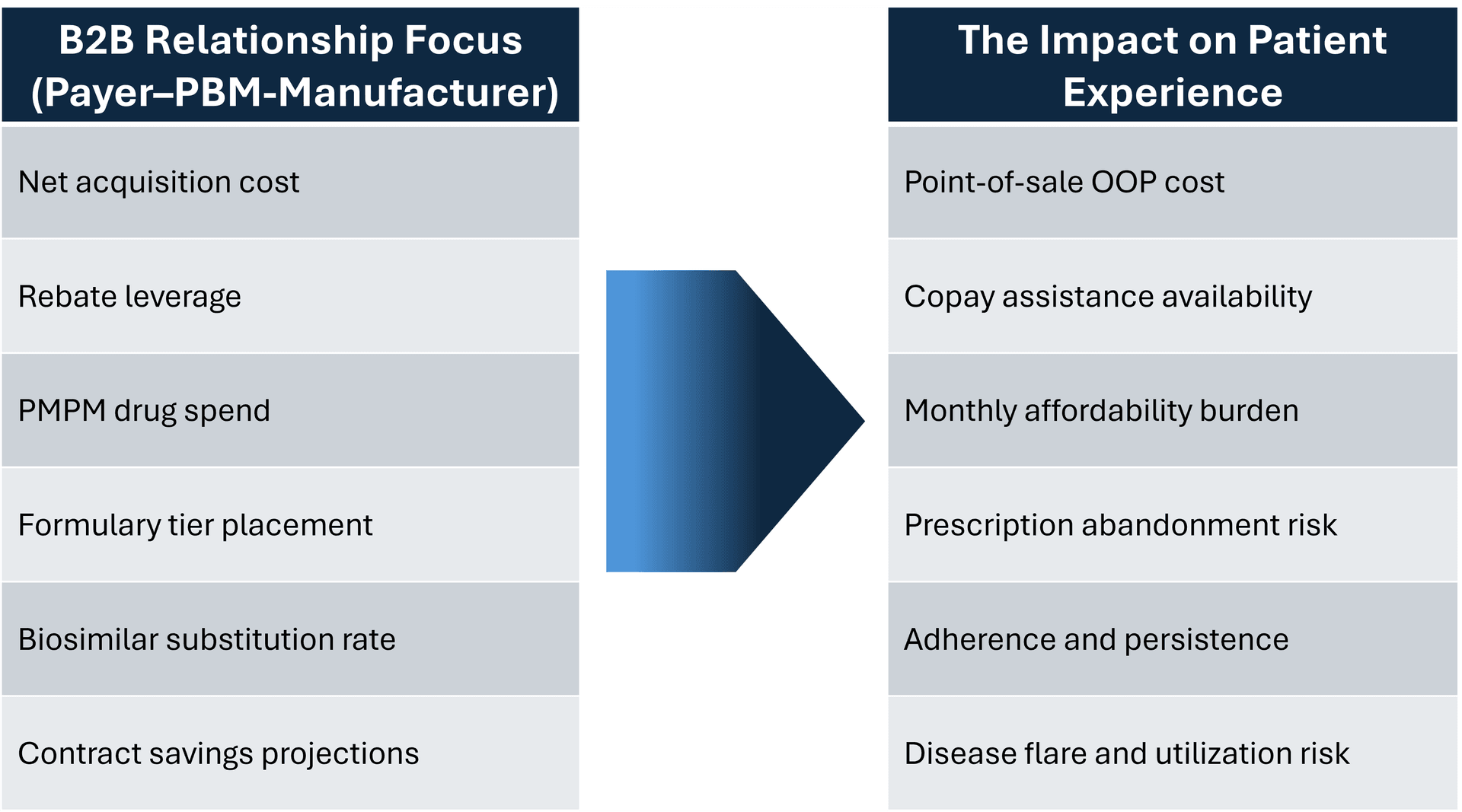
The biosimilar debate is largely framed as a transaction between manufacturers, PBMs, and health plans. The language centers on acquisition cost, rebate leverage, and projected PMPM savings. It is a business-to-business optimization exercise.
What is notably absent is the patient.
Specifically, absent from the discussion is the evidence on what keeps patients stable, adherent, and out of higher-cost settings. There is no lack of data showing that point-of-sale affordability drives adherence. Copay assistance, predictable cost exposure, and financial continuity materially influence persistence in chronic disease. Yet those variables rarely appear in formulary deliberations.
The commercial negotiation optimizes net price.
The patient experiences [suboptimal] benefit design.
The table below illustrates that misalignment. On one side is the B2B focus driving contracting decisions. On the other is the patient-facing reality that determines whether therapy is taken consistently enough to deliver value.
If a formulary decision lowers net payer cost but increases abandonment or destabilizes disease control, system savings may be offset by downstream medical costs.
Net price is not the same as patient price.
That distinction should be central in biosimilar policy discussions and likely even more broadly.
Policy and Benefit Design Solutions to Improve Biosimilar Affordability
To align biosimilar competition with patient affordability:
- Protect point-of-sale affordability for high-value chronic biologics
- Evaluate copay accumulator policies using real-world adherence data
- Incorporate abandonment and adherence metrics into outcomes-based contracts
- Apply value-based insurance design principles to specialty medications
Biosimilars are a critical tool for controlling drug spending, but competition that increases financial toxicity undermines value.
Lower acquisition cost alone is not the goal. Lower total cost of care, without shifting burden to patients, should be.
References:
1. Brixner D, Rubin DT, Mease P, et al. Patient support program increased medication adherence with lower total health care costs despite increased drug spending. J Manag Care Spec Pharm. 2019.
2. Lalani HS, Hwang CS, Kesselheim AS, Rome BN. Strategies to help patients navigate high prescription drug costs. JAMA. 2024.
3. Feng K, Russo M, Maini L, Kesselheim AS, Rome BN. Patient out-of-pocket costs for biologic drugs after biosimilar competition. JAMA Health Forum. 2024.
4. Feng K, Kesselheim AS, Russo M, Rome BN. Patient out-of-pocket costs following the availability of biosimilar versions of infliximab. Clin Pharmacol Ther. 2022.
5. Sherman BW, Epstein AJ, Meissner B, Mittal M. Impact of a co-pay accumulator adjustment program on specialty drug adherence. Am J Manag Care. 2019.
6. Patel A, Sheinson D, Wong WB. Patient liability, treatment adherence, and treatment persistence associated with state bans of copay accumulator adjustment programs. J Manag Care Spec Pharm. 2024.
7. Hopson S, Saverno K, Liu LZ, et al. Impact of out-of-pocket costs on prescription fills among new initiators of biologic therapies for rheumatoid arthritis. J Manag Care Spec Pharm. 2016.
8. Wong WB, Donahue A, Thiesen J, Yeaw J. Copay assistance use and prescription abandonment across race, ethnicity, or household income levels for select rheumatoid arthritis and oral oncolytic medicines. J Manag Care Spec Pharm. 2023.
9. Harrold LR, Briesacher BA, Peterson D, et al. Cost-related medication nonadherence in older patients with rheumatoid arthritis. J Rheumatol. 2013.
10. Riegler JS, Kesselheim AS, Rome BN. Out-of-pocket spending for biologic drugs after biosimilar competition for Medicare patients. JAMA Netw Open. 2026.
11. Chandra A, Flack E, Obermeyer Z. The Health Costs of Cost-Sharing. NBER Working Paper 28439. 2021.
12. Faridi KF, Dayoub EJ, Ross JS, et al. Medicare Coverage and Out-of-Pocket Costs of Quadruple Drug Therapy for Heart Failure. J Am Coll Cardiol. 2022 Jun 28;79(25):2516-2525.
